This will seek to optimise supply chains for the pharma industry as a whole – by introducing a data-driven approach offering traceability and operational efficiency. In view of the globalisation of pharma supply chains, NE is setting up Good Distribution Practices (GDP) -compliant business locations in major markets worldwide to enhance its supply networks on a global level. This should enable safer and more secure supply of pharmaceuticals.
Nippon Express background
The “Nippon Express Group Management Plan 2023 — Dynamic Growth” says the Company must strengthen its business approach. In determining how to tailor services to the needs of individual customers, NE plans to focus efforts on a customer/industry-based approach which seeks to see services from the customer’s perspective.
To ensure it has in place the extensive expertise and strict quality control required for pharmaceutical logistics, NE has created:
- a Pharmaceuticals Business Division
- a Pharmaceuticals Logistics Quality Assurance Office.
These establish uniform group-wide standards across:
- operational frameworks
- human resource development
- quality assurance regimes
- etc.
Increasing supply chain sophistication by being data-driven
Earlier in 2020 Nippon Express established a Digital Platform Strategy Office. The objective is to provide the pharmaceutical industry (and other industries) with solutions designed to optimise supply chains.
To this end, construction is underway of a digital platform for the pharmaceutical industry that will:
- exploit IoT devices and blockchain technology
- link temperature control and other logistics information end-to-end
- make use of this logistics information to develop commercial distribution services.
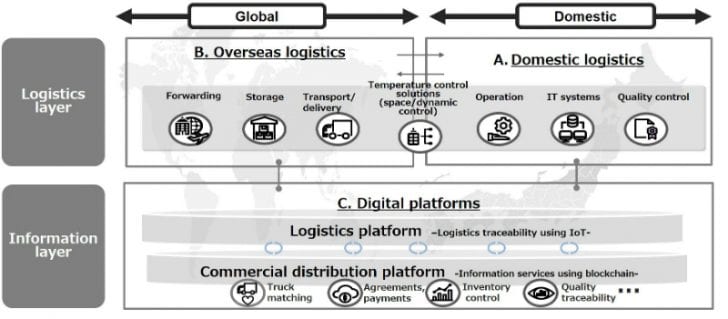
Constructing supply networks – in Japan and globally
Nippon Express is already setting up pharmaceutical-specific facilities in East Japan, West Japan, Kyushu and Toyama. These will complement the existing ‘medical hubs’ located at the Narita and Kansai international airports (which have long had involvement in the import/export of raw materials and finished products).
All these facilities will gain the equipment to handle GDP-compliant quality control and business continuity plans relevant to pharmaceutical supply. Platforms will be constructed to enable joint warehouse storage and transport. Operations previously carried out on an individual customer basis will deliver greater operational efficiency.
Nippon Express will also draw on its global network. It will link infrastructure and forwarding functions and develop overseas logistics networks. Again, NE’s objective is to that guarantee safety on a global level as required for pharmaceutical logistics. More specifically it has already opened GDP-certified CFS in:
- London and Milan, which both host clusters of manufacturers of new drugs
- India, where generics are driving robust market growth
- the U.S., which remains the world’s largest market.
Enterprise Times: what does this mean
Prima facie, Nippon Express’ actions are unsurprising. Long tail supply chains and blockchain are coming together, albeit more slowly than many expected (and hoped).
Where NE may ‘score’ is that it will be able to make full use of and build on its existing global pharmaceutical supply networks. These already serve pharma customers. Adding blockchain/IoT in the pharmaceutical industry and create new value through logistics. NE also indicates it is ready to enhance its network through M&A deals as well as other means – with an eye to offering storage and distribution logistics in major markets.